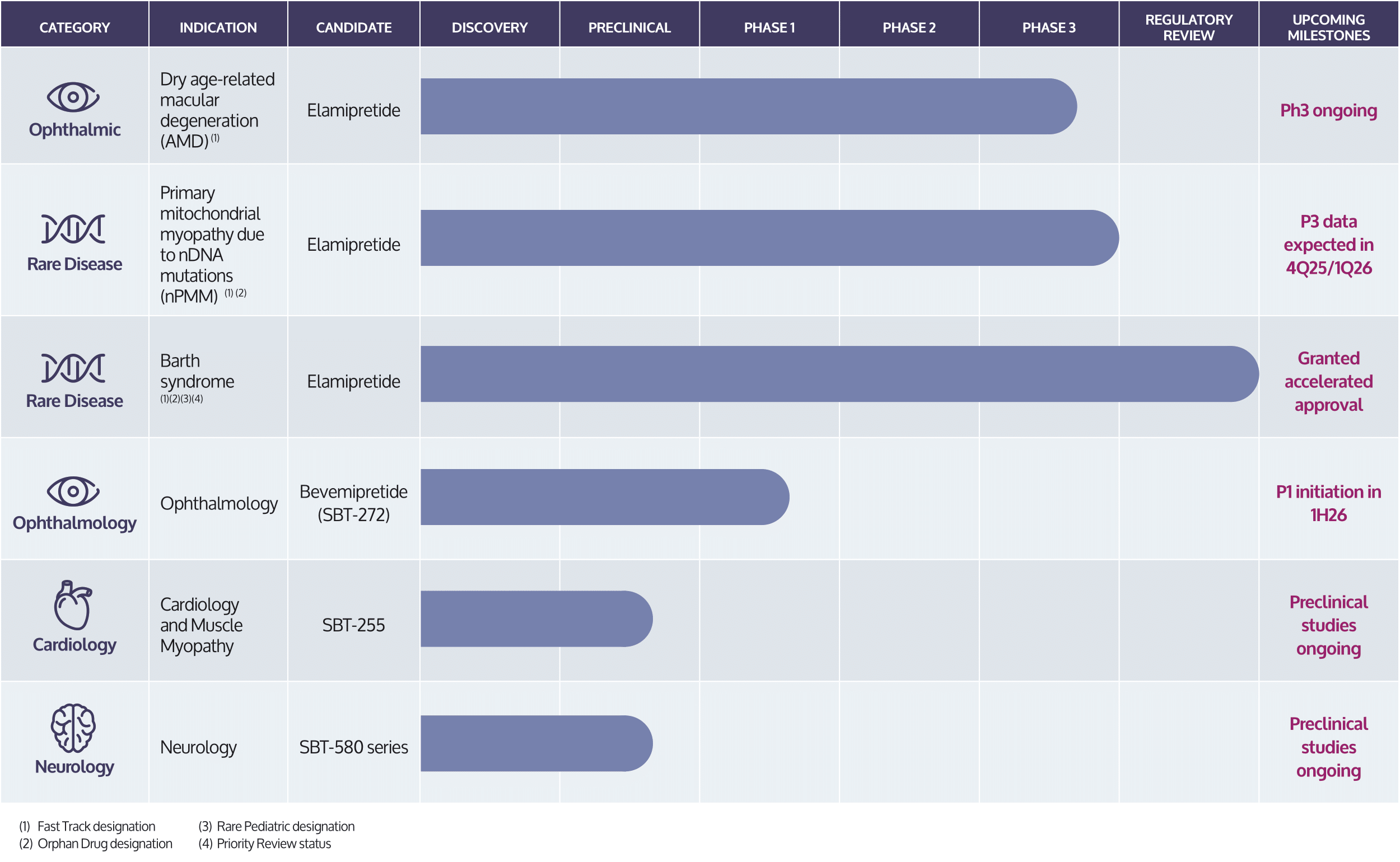
Stealth’s lead investigational product candidate, elamipretide, is a peptide compound that readily penetrates cell membranes, and targets the inner mitochondrial membrane where it binds reversibly to cardiolipin.13 In preclinical or clinical studies, we have observed that elamipretide increases mitochondrial respiration, improves electron transport chain function and ATP production and reduces formation of pathogenic ROS levels.13-19 This elamipretide-cardiolipin association has been shown to normalize the structure of the inner mitochondrial membrane, thereby improving mitochondrial function.13 Functional benefit is achieved through improvement of ATP production and interruption and potential reversal of damaging oxidative stress.13 In September 2025, the FDA granted accelerated approval of elamipretide, which is being marketed in the U.S. under the trade name FORZINITYTM (injection), a mitochondrial cardiolipin binder indicated to improve muscle strength in adult and pediatric patients with Barth syndrome weighing at least 30 kg. FORZINITY is the first ever treatment option for Barth syndrome and the first approved mitochondria-targeted therapeutic. We are investigating elamipretide in late-stage clinical studies in ophthalmic diseases entailing mitochondrial dysfunction, such as dry AMD, rare neuromuscular disorders, such as primary mitochondrial myopathy. We are evaluating our second-generation clinical-stage candidate, Bevemipretide (SBT-272), for rare neurological disease indications, such as amyotrophic lateral sclerosis, and have a deep pipeline of novel compounds under evaluation for rare neurological and cardiac disease indications.
Healthy & Unhealthy